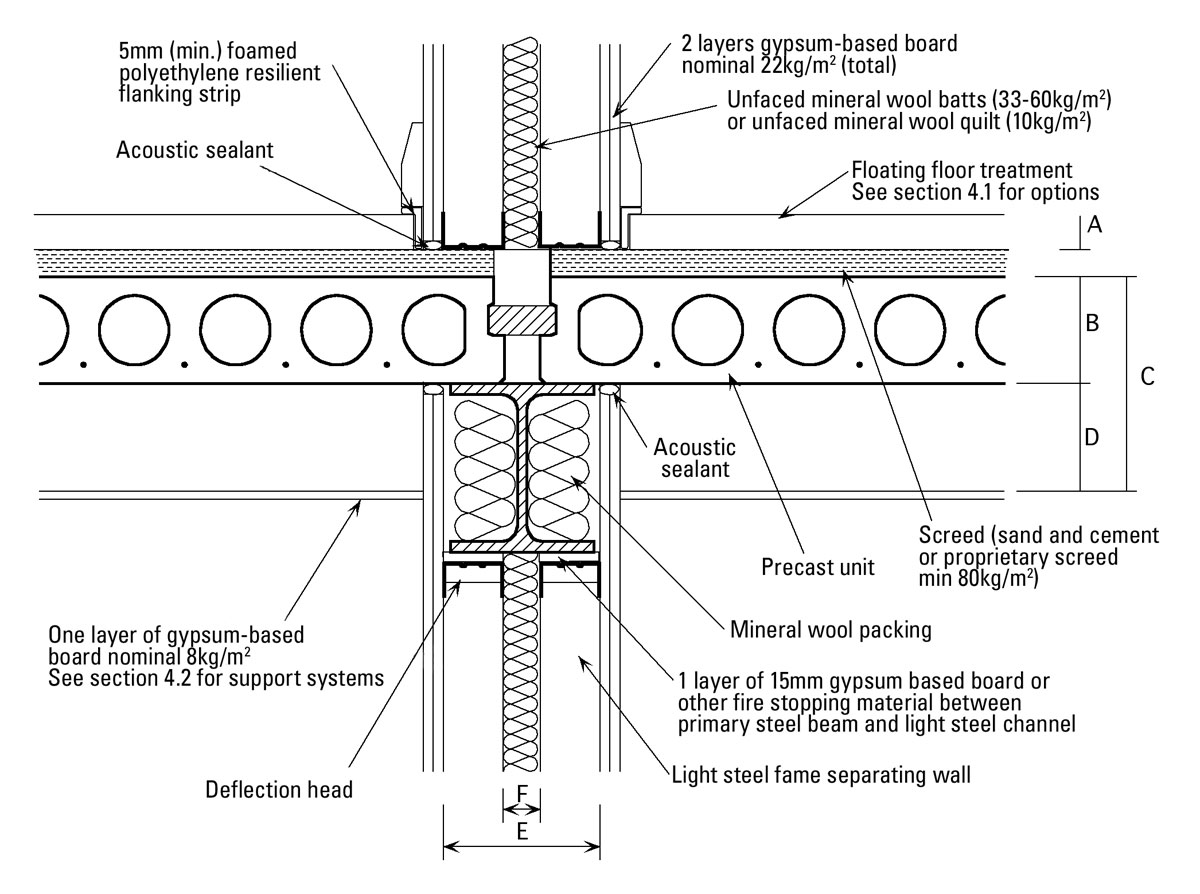
Technical
AD 339: Stainless steel in contact with other metals
Advisory Desk Note AD338 gave a summary of sources for guidance on the use of stainless steel. One of the publications referred to, Stainless Steel in Contact with Other Metallic Materials, published by Euro Inox, addresses the issue of galvanic (bimetallic) corrosion. This Advisory Desk Note summarizes the guidance given in that publication and also the recommendations in the Design Manual for Structural Stainless Steel.
Principles of galvanic corrosion
When two dissimilar metals are in contact and also bridged by an electrolyte (i.e. an electrically conducting liquid), a current flows from the anodic metal to the cathodic or nobler metal through the electrolyte. As a result, the less noble metal may corrode at a rate far higher than would be expected if it were not in contact with the noble metal. The more noble material (the cathode) is protected against corrosion. In some cases, this can lead to corrosion in metals that would otherwise be corrosion resistant in the environment in question. Seawater is a strong electrolyte; however, rain or humidity may be enough to trigger a galvanic reaction.
Stainless steels usually form the cathode in a bimetallic couple and therefore do not suffer corrosion. Contact between austenitic stainless steels and aluminium may result in some additional corrosion of the latter metal. This is unlikely to be significant structurally, but the resulting white/grey powder may be deemed unsightly. Galvanized steel in contact with stainless steels is not normally considered to be a serious corrosion risk, except possibly in severe (marine type) environments. Contact with copper should generally be avoided. When welding stainless steel to carbon or low alloy steel, it is important to select welding consumables that are at least as noble as the parent material.
Parameters which affect the risk of galvanic corrosion
The risk of galvanic corrosion occurring is difficult to predict as it depends on numerous factors. Apart from the metals themselves, environment and design are crucially important. The main factors are addressed below.
Potential difference
The potential difference between the two metals alone is not a good indicator of the actual risk of galvanic corrosion. The important factor is not the difference of potential observed under standardized experimental conditions but rather the actual difference of potential under real conditions (where the presence of surface oxide films, effects of area ratios and different electrolyte chemistry all have an effect).
Conductivity of electrolyte
The risk of galvanic corrosion increases with increasing electrolyte conductivity. Water film resulting from condensation would be an example of an electrolyte with low conductivity while sea water is an electrolyte with high conductivity.
Wetting duration and environments
There is a strong interaction between electrolyte conductivity and duration of wetting. This is of critical importance wherever components are not permanently wetted. Both length of exposure and electrolyte conductivity are strongly dependent on local conditions. In marine, industrial or indoor swimming pool environments, the risk of galvanic corrosion is significantly higher than in rural atmospheric conditions. Besides ambient atmosphere, design details play a decisive role. Factors that help humidity films to dry quickly (adequate aeration, prevention of crevices, free drainage of rainwater) reduce corrosive attack. Permanently humid areas in crevices or covered areas, stagnant water and solid surfaces can considerably accelerate galvanic corrosion.
Cathode and anode area
The risk of attack is greatest if the area of the more noble metal (e.g. stainless steel) is large compared with the area of the less noble metal (e.g. carbon steel). Adverse area ratios are likely to occur with fasteners and at joints. Carbon steel bolts in stainless steel members should be avoided because the ratio of the area of the stainless steel to the carbon steel is large and the bolts will be subject to aggressive attack. Conversely, the risk of attack of a carbon steel member by a stainless steel bolt is negligible. It is usually helpful to draw on previous experience in similar sites because dissimilar metals can often be safely coupled under conditions of occasional condensation or dampness with no adverse effects, especially when the conductivity of the electrolyte is low.
Preventing galvanic (bimetallic) corrosion
Galvanic corrosion is rarely a problem in building environments, although it is generally accepted as good practice to take the preventative measure of inhibiting current flow between the dissimilar metals. There are two means to achieve this:
- insulating the dissimilar metals, i.e. breaking the metallic path,
- preventing electrolyte bridging by breaking the electrolytic path, by applying paint or other coating. Where protection is sought by this means and it is impracticable to coat both metals, then, surprisingly, it is preferable to coat the more noble metal (i.e. coat the stainless steel, in the case of a stainless steel / carbon steel connection)
Further information:
Stainless steel in contact with other metallic materials. Materials and Applications Series, Volume 10, Euro Inox, 2009. Available from www.euro-inox.org
Design Manual for Structural Stainless Steel. Euro Inox and the Steel Construction Institute, Third Edition, 2006. Available from www.euro-inox.org and www.steel-stainless.org/designmanual
PD 6484:1979 Commentary on corrosion at bimetallic contacts and its alleviation. (This PD documents the general behaviour of metals in bimetallic contact in rural, urban, industrial and coastal environments.)
BSSA provide technical information on this subject on their web site www.bssa.org.uk and they also run a Stainless Steel Advisory Service for technical queries (Telephone 0114 2671265, Email ssas@bssa.org.uk). BSSA offer a range of Continuing Professional Development modules on the use of stainless steel in construction for in-house training.
Contact: Nancy Baddoo
Tel: 01344 636525
Email: advisory@steel-sci.com